Air Pollution Control Plant
Air control Engineering Co., Ltd.
Chemical Adsorption
Understanding Chemical Adsorption
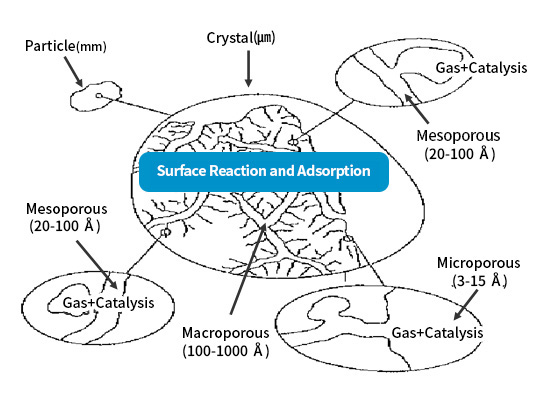
- Chemical adsorbents are products made by attaching or mixing reagents that efficiently react with the target gas to a porous material.
- The porous product facilitates the physical adsorption of the target gas (capillary action), maximizing the removal rate of the reagent. The reagent reacts with the target gas and becomes fixed within the porous pores after the reaction.
- Chemical adsorbents can be classified into organic and inorganic reagents.
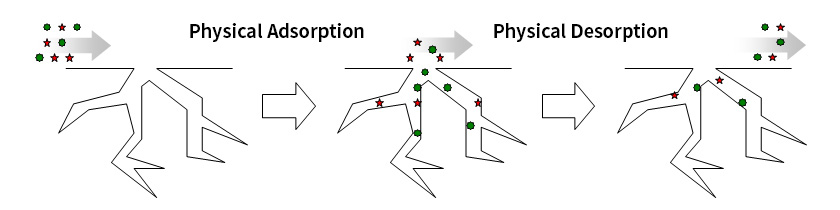
Principle of Physical Adsorption Removal
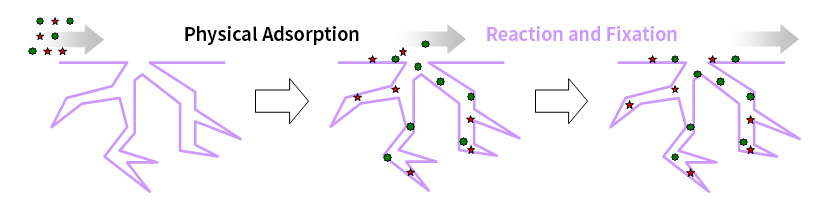
Principle of Chemical Adsorption Removal
Comparison of Physical Adsorption vs. Chemical Adsorption
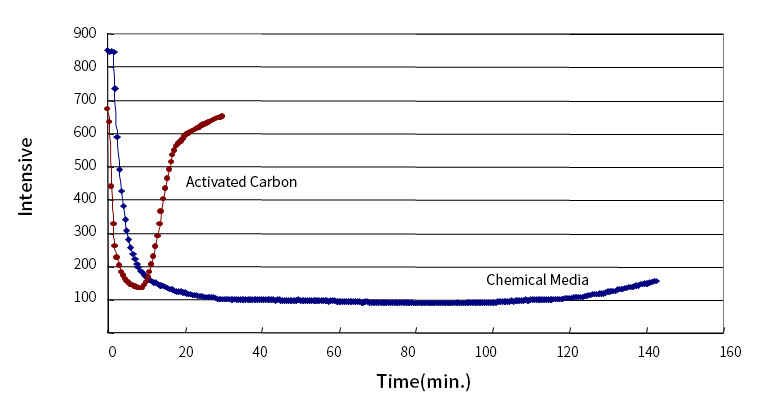
Adsorption Performance Evaluation [TMA; Trimethylamine]
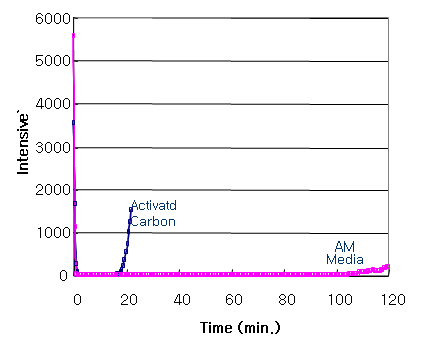
Adsorption Performance Evaluation – Mercaptane
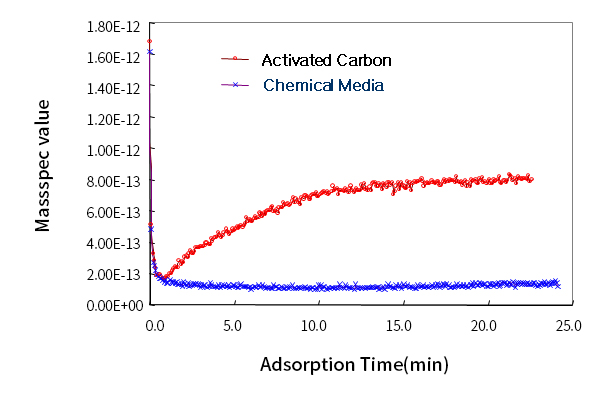
Tenfold Adsorption Performance of General Activated Carbon
Types of Chemical Media

- MEDIA FOR SULFURIC GAS
- MEDIA FOR AMINE GAS
- MEDIA FOR ALDEHYDE GAS
- MEDIA FOR VOC GAS
- MEDIA FOR ACID GAS
- MEDIA FOR TOXIC GAS
- MEDIA FOR Influenza Virus & AI Virus
Table : Chemical Media List
| Classification | Organic Media | Inorganic Media | Special | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Target Gas | CN | CS | CM | CX | CD | AM | ICA | AO | ST |
| NH3 | ● | ○ | |||||||
| SOx | ● | ● | |||||||
| NOx | ● | ● | |||||||
| O3 | ● | ||||||||
| Cl2 | ● | ● | |||||||
| HF | ● | ● | |||||||
| HCl | ● | ● | |||||||
| H2S | ● | ● | ● | ||||||
| MM | ● | ||||||||
| TMA | ● | ||||||||
| Ethylene | ● | ● | |||||||
| CO2 | ● | ||||||||
| CO | ● | ||||||||
| Aldehyde | ● | ||||||||
| PGMEA | ● | ||||||||
| PGME | ● | ||||||||
| Phosgene | ● | ||||||||
| Acetic Acid | ● | ○ | ○ | ||||||
| PFC | |||||||||
| VOC | ○ | ○ | ○ | ○ | ○ | ● | ○ | ||
| Virus | ● | ||||||||
ACESORB-CN

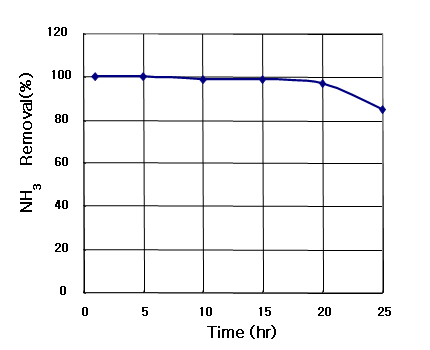
NH3 Conc.: 50 ppm, S.V= 1,200 min-1,
RH = 50%, Mass Analyzer
Product information
| Composition | Chemical impregnated carbon | |
|---|---|---|
| Particle shape | Extrudated | Crushed |
| Target contaminants | NH3, TMA, Amine, Amide | |
| Use | Filling in case or coating on supported material | |
| Application | Living room, Toilet, Office | |
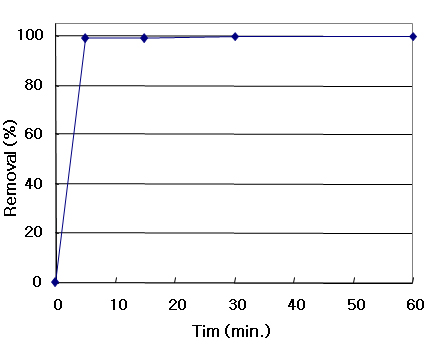
ACESORB-AM

SO2 Conc.: 200 ppm, S.V= 3,6000 hr-1,
RH = 50%, Gas Sensor
Product information
| Composition | Chemical impregnated metal oxide | |
|---|---|---|
| Particle shape | Sphere | Extrudated |
| Target contaminants | Sulfur compounds, NOx, Ethylene | |
| Use | Filling in casel | |
ACESORB-CS

Acetic acid Conc.: 1㎕, Chamber Test
RH = 50%, Chemical test kit
Product information
| Composition | Chemical impregnated carbon | |
|---|---|---|
| Particle shape | Extrudated | Crushed |
| Target contaminants | SOx & NOx, H2S, Acid compounds | |
| Use | Filling in case or coating on supported material | |
ACESORB-CM

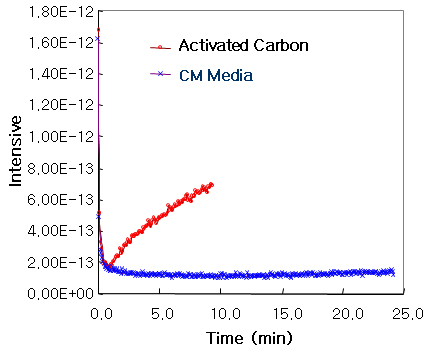
Methyl Mercaptan Conc.: 500 ppm,
Flow Rate 100cc/min, Mass Analyzer
Product information
| Composition | Chemical impregnated carbon | |
|---|---|---|
| Particle shape | Extrudated | Crushed |
| Target contaminants | Sulfur compounds, Organic acids, Phosgene etc. | |
| Use | Filling in case or coating on supported material | |
| Application | Refrigerator, Kitchen | |
ACESORB-ICA

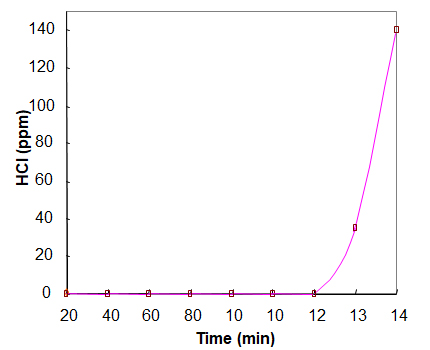
HCl Conc.: 10,000 ppm,
Flow Rate 650cc/min, Mass Analyzer
Product information
| Composition | Metal oxide mixture |
|---|---|
| Particle shape | Extrudated(Crushed) |
| Target contaminants | Chlorine, Hydrochloric acid, Carbon dioxide |
| Use | Swimming pools, PVC molding utility |
ACESORB-CX

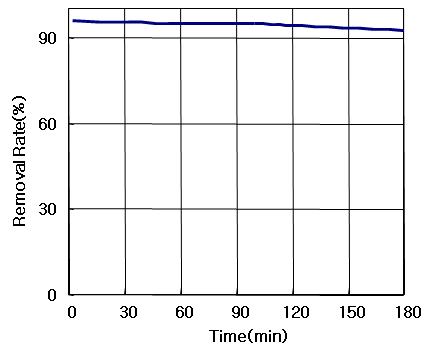
Ozone Conc.: 2,000 ppm,
Flow Rate 2,000cc/min, Mass Analyzer
Product information
| Composition | Chemical impregnated carbon | |
|---|---|---|
| Particle shape | Extrudated | Crushed |
| Target contaminants | VOCs, Ozone, Hydrocarbons | |
| Use | Filling in case or coating on supported material | |
| Application | Kitchen, Electric Heater | |
ACESORB-CD

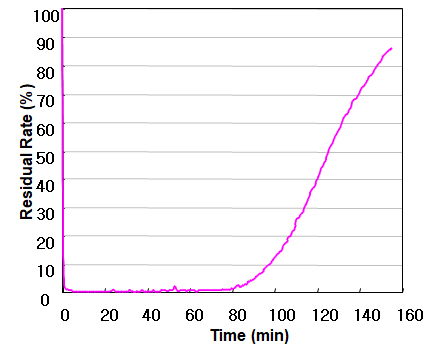
Acetaldehyde Conc.: 330 ppm,
Flow Rate 340cc/min, Mass Analyzer
Product information
| Composition | Chemical impregnated carbon | |
|---|---|---|
| Particle shape | Extrudated | Crushed |
| Target contaminants | Acetaldehyde, Formaldehyde, Acetic acid | |
| Use | Filling in case or coating on supported material | |
ACESORB-AO

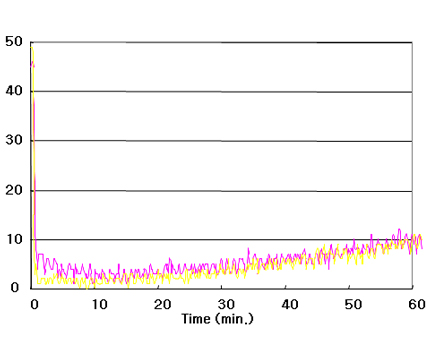
Carbon Mono-oxide Conc.: 50 ppm,
Flow Rate 1,000cc/min, Mass spec.
Product information
| Composition | Chemical impregnated metal oxide |
|---|---|
| Particle shape | Sphere |
| Target contaminants | Carbon mono-oxide, Aldehydes |
| Use | Filling in case |
ACSEORB-ST

KTR의 “Sterilization Efficacy Assessment”
- 1Staphylococcus Aureus (Staphylococcus Aureus)
- 2Klebsiella Pheumoniae => Evaluation Result: 99.9% Sterilization Efficacy

Ref. Analyzer for Deodorant
Test Utility Ⅰ
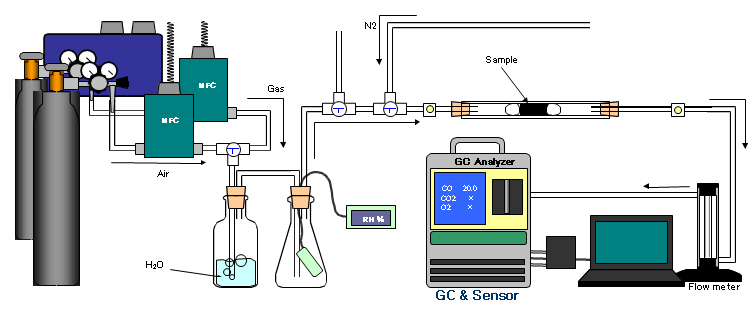
Real Application Case
Chemical Adsorption Reaction Equations for Toxic Gas Storage Facilities
| ROOM NO | Classification | GAS NAME | CHEMICAL ADSORPTION | CHEMICALL ADSORBENTS | Reaction Equations |
|---|---|---|---|---|---|
| R-1 | Toxic Gas Storage 1 | ASH2 | B-1 | 4AsH2 + 11CuO → 2Cu2As + As2O5 + 7Cu + 6H2O | |
| SiH4 | MTx | SiH4 + 2MOH →àM2Si + 2H2O + H2 | |||
| Si2H6 | CARBON | Si2H6 + 4MOH →à2M2Si + 4H2O + H2 | |||
| B2H6 | B2H6 + 3CuO → B2O3 + 3Cu+3H2 | ||||
| PH3 | PH3 + 3CuO → Cu3P + P + 3H2O | ||||
| R-2 | Toxic Gas Storage 2 | ASH2 | B-2 | ||
| NH3 | CN | NH3 + MeSOx →à(NH3)nSOx | |||
| R-3 | Toxic Gas Storage 3 | C2H4O | B-3 | C2H4O --> Adsorption Removal | |
| CO | CE | CO + O2 → CO2 | |||
| HC | CARBON | HC ---> Adsorption Removal | |||
| R-4 | Toxic Gas Storage 4 | HCI | B-4 | MF | 3HCl + MO(OH) →àMCl3 + 2H2O |
| NF3 | CARBON | NF3 : NF3 + A/C → Adsorption Removal | |||
| CI2 | CS | Cl2 + 2MOH → 2MCl + H2O + 1/2O2 Cl2 + M(OH)2 → MCl2 + H2O + 1/2O2 |
|||
| R-5 | Toxic Gas Storage 5 | NO | B-5 | CARBON | AC + NO → CN*, CNO*, CNO3* |
| so2 | CX | MeOH + SO2 → MeSOx + H2O |


Physical Adsorption
ACTIVATED CARBON TOWER
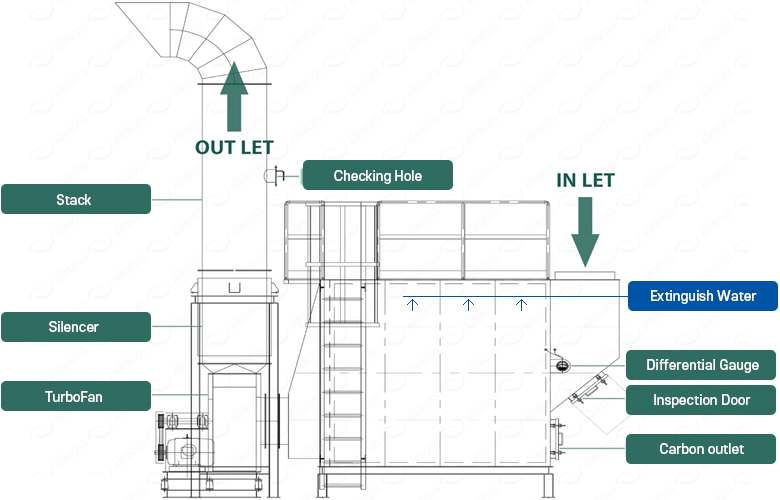

PROCESS DESCRIPTION
An Activated Carbon Tower is a facility designed to absorb and adsorb various organic gases and odors through physical and chemical methods. It removes substances based on the free bonding of atoms on the surface of activated carbon and the chemical bonding strength depending on gas properties. It is applicable for most low-concentration gases, and by using specific adsorbents, target substances can be selectively removed, making it widely used in industrial applications and environmental protection systems. Adsorbents (such as activated carbon and silica gel) are porous, and the greater the number of capillaries and the higher the affinity for the adsorbed substances, the greater the adsorption effect.
Types and Uses of Adsorbents
| Adsorbent | Use |
|---|---|
| Activated Carbon | Solvent recovery, odor removal, gas purification (most widely used) |
| Alumina | Drying gases, air, and liquids |
| Bauxite | Removal of oil fractions, drying gases and solutions |
| Bone Char | Decolorization of sugar |
| Decoloring Carbon | Removal of oil, pigments, fractions, and waxes; decolorization of beverages |
| Fuller's Earth | Purification of lubricating oils and fatty waxes |
| Magnesia | Purification of gasoline and fatty waxes |
| Silica Gel | Removal of impurities in caustic soda solutions |
| Strontium Sulfate | Drying and purification of gases, removal of iron in caustic soda solutions |
Types and Characteristics of Activated Carbon

Figure3. constructed carbon

Figure4. granular active carbon

Figure5. powdered active carbon
- Constructed Carbon is expensive but shows high adsorption rates, primarily used for treating high-concentration gases.
- Granular or Pelletized Activated Carbon has a sufficiently large surface area and experiences low pressure drop in fixed bed applications, making it relatively easy to recover adsorbed substances. It is the most widely used type.
- Powdered Activated Carbon is inexpensive but of lower quality than granular carbon. It has significant limitations due to high pressure drop when used in packed towers. Additionally, powdered carbon is nearly impossible to regenerate and must be disposed of after use.
Microscopic Images of Granular Activated Carbon Pores
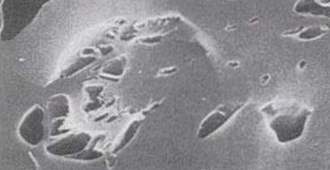
Figure6. Granular carbon (x3000)

Figure7. Peller type active carbon (x3000)
| Bulk Density | 22-34lb/ft² |
|---|---|
| Hear Capacity | 0.27-0.36Btu-lb℉ |
| Pore Volume | 0.56-1.20㎤ / g |
| Surface Area | 600-1600㎠ / g |
| Average Pore Diameter | 15-25 |
| Regeneration Temperature:steam | 100-140℃ |
| Maximum Allowable Temp | 150℃ |
Adsorption Capacity of Activated Carbon by Odor Components

Operation of Adsorption Tower
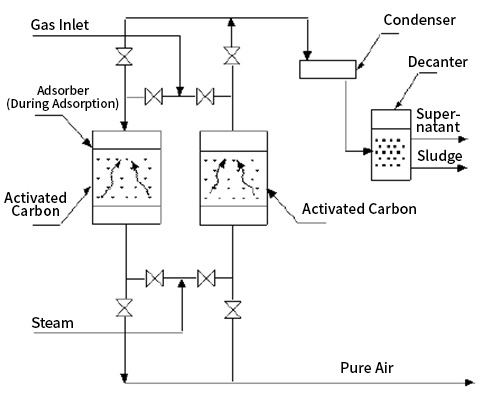
Regeneration of the adsorbent by desorption can be achieved through various methods, depending on the properties of the adsorbent. Typically, when desorbing gas or vapor, the temperature of the adsorbent-adsorbate is increased, often using a pressure cycle (thermal swing). A common thermal swing method involves passing hot gas through the adsorbent layer, cooling the outgoing gas, and recovering it during the separation process.
Types of Activated Carbon Towers
1. Fixed Bed Adsorber
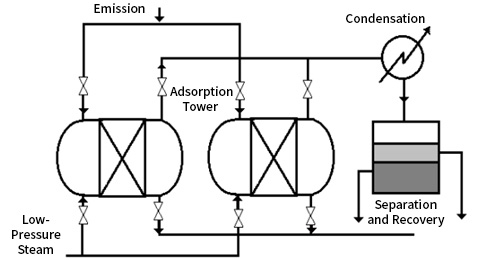
2. Continuous Adsorber (Moving Bed Adsorber)
3. Fluidizing Adsorber
Breakthrough Point
At this point, the vapor that should be adsorbed appears in the outlet gas, and it is said that the activated carbon is saturated when the concentrations of vapor at the inlet and outlet are equal. (Refer to Figure 8: Breakthrough Curve)
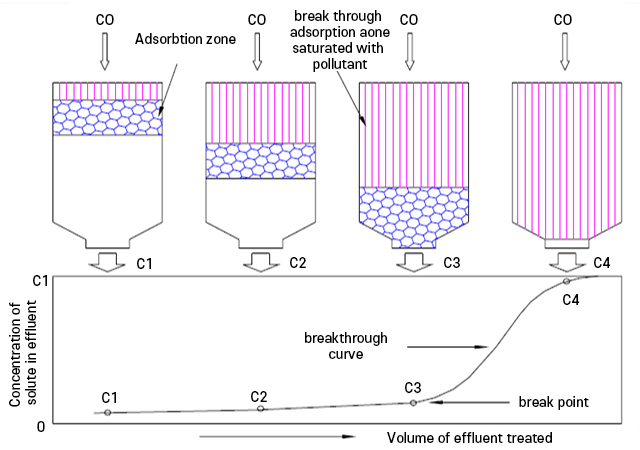
Figure 8. breakthrough curve
Fire Prevention Technologies for Adsorption Towers
- 1The ignition temperature for palm shell activated carbon is about 200°C, while for coal-based activated carbon it is approximately 350°C. Therefore, coal-based activated carbon is more favorable. To avoid heat accumulation, use uniformly shaped constructed activated carbon.
- 2If dead zones exist, heat accumulation can occur. The structure of the activated carbon layer should be vertical or inclined, and the thickness (height) of the activated carbon layer should be installed to be 0.5 m or less.
- 3Maintain a contact time of less than 2 seconds, which corresponds to a linear velocity of 0.2 m to 0.4 m/sec. If the linear velocity is below 0.2 m/sec, there is a potential for heat accumulation.
- 4Install a wet scrubber or heat exchanger at the front of the adsorption tower, or dilute with air to maintain the temperature below 70°C.
- 5During the initial operation, adsorption heat is generated, which gradually decreases after 15 to 30 minutes. Sufficient water should be sprayed, and the system should be run in idle for about 30 minutes before normal operation. The water added initially will naturally desorb during operation without reducing the adsorption capacity of the activated carbon.
- 6Install thermocouples and a temperature detection alarm system in the adsorption tower, with a set point of 100°C (adjustable based on operating conditions). A safety mechanism should be in place to activate water spray if the temperature rises.
- 7During shutdown, switch the incoming gas to cooler air and run the blower in idle for about 30 minutes to lower the internal temperature of the adsorption tower to below 50°C before completing the shutdown.
